“Fat burns in a carbohydrate flame.”
Maybe you’ve heard it before, but what exactly does it mean? What implications does it have on health and weight control?
This expression has been resurrected lately with the resurgence in popularity of ketogenic diets (aka, low-carbohydrate, low-carb diets) and their role in ketosis.
I’ll discuss these in context below. [also check out my related article on carbohysteria]
If you’ve ever taken a university biochemistry class then maybe you can still recall some of this through a distant haze of pot smoke and hangovers (if you’re a university student reading this now, put down that joint!).
If you’re just a health enthusiast and curious to understand this carbohydrate flame thing a little bit better, then pay close attention and I’ll try to keep this as logical and understandable as possible.
Disclaimer: physiology and biochemistry are complex, so I have only given an extremely basic overview of metabolic pathways and cellular processes as it applies to answering the carb flame question.
“Carbohydrate flame” backstory
Before you can understand the “carbohydrate flame,” it’s important to set the stage by discussing two metabolic conditions:
- Normal, well fed conditions (i.e., 24 hour supermarkets, fast food on every corner).
- No-carb or low-carb intake, fasting, starvation conditions (i.e., you’re Tom Hanks stranded on a South Pacific island – WILSON!!).
Normal, well-fed conditions
Under normal conditions, you rely primarily on three energy-containing nutrients:
- Carbohydrate
- Fat
- Protein
In digestion, carbohydrate is broken down into glucose, protein to amino acids, and fat to fatty acids.
These are absorbed and taken up to be stored for later or burnt now for energy.
Macronutrient |
Broken down to |
|---|---|
| Carbohydrate | Glucose |
| Fat | Fatty acids |
| Protein | Amino acids |
Carbohydrate is your central nervous system’s preferred high-octane fuel (i.e., your brain and nerves). Got it? Remember this because you’ll see it again.
Your body stores carbohydrate in the liver and muscles (called glycogen) and it can be broken down at rest (i.e., liver glycogen to maintain blood sugar) or in times of need (i.e., muscle glycogen to fuel exercise, slay the saber-toothed tiger, etc).
Fat is also used as a fuel source at rest and during prolonged exercise, as is protein to a lesser extent under certain metabolic conditions.
Energy producing metabolic pathways
There are two energy pathways by which your body liberates energy from the foods you eat:
- Glycolysis
- Krebs cycle (also called the tricarboxylic acid cycle or TCA cycle)
Glycolysis is the pathway you use to break down carbohydrate.
The Krebs cycle is the pathway you use to fully oxidise (burn) glucose and fatty acids down to carbon dioxide and water (and amino acids, but not as a main fuel source).
For the purists out there, yes, I know oxidative phosphorylation in the electron transport chain also generates a lot of ATP, but I’m focusing on the Krebs cycle for simplicity’s sake.
These cycles both liberate the chemical energy stored in food and produce adenosine triphosphate (ATP), which is like your body’s chemical “money” it needs to “pay” for different chemical and physiological “transactions” (i.e., muscle contraction, cellular reactions, etc).
Glycolysis
Glycolysis translates to “glyco” which means glucose (or sugar) and “lysis” which means splitting or breaking down, so it’s a sugar-splitting or sugar-breaking down process. Note: you do not burn fat in the glycolytic pathway.
One molecule of glucose contains six carbons. It goes through glycolysis and is broken down into two 3-carbon molecules of pyruvate.
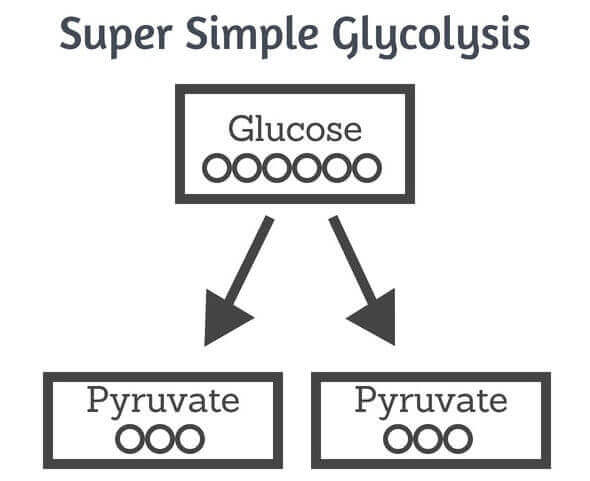
To completely oxidise glucose, pyruvate is transferred inside the mitochondria where it is further broken down to a 2-carbon molecule called acetyl coenzyme A (acetyl coA) – the other carbon is flicked off in carbon dioxide.
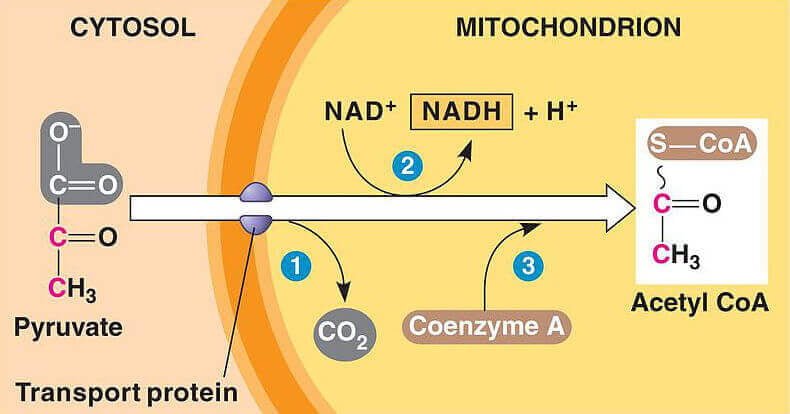
Acetyl coA then enters the Krebs cycle, combining with oxaloacetate to form citrate, and eventually passing through the entire cycle, down through the electron transport chain where it can be broken down into carbon dioxide and water.
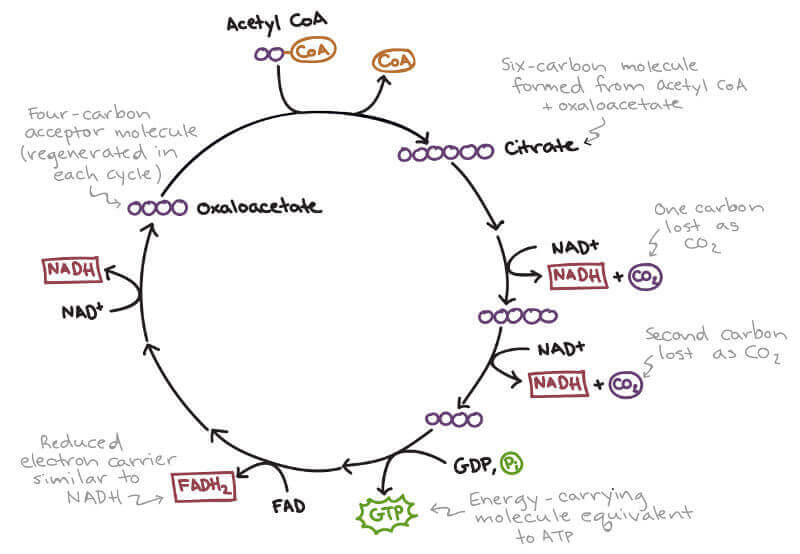
Recall from above, the entire process generates energy in the form of ATP which is like a “chemical currency” to “pay” for different metabolic “transactions” such as muscle contraction.
Krebs cycle for fat oxidation
What about burning fat? How does that work?
Enter the Krebs cycle.
Your body stores fat as triglycerides and can break them down for energy (such as when you go for a long run or bike ride).
The name “triglyceride” refers to fat’s chemical structure. A triglyceride has three (tri) fatty acids connected to a glycerol (glyceride) molecule.
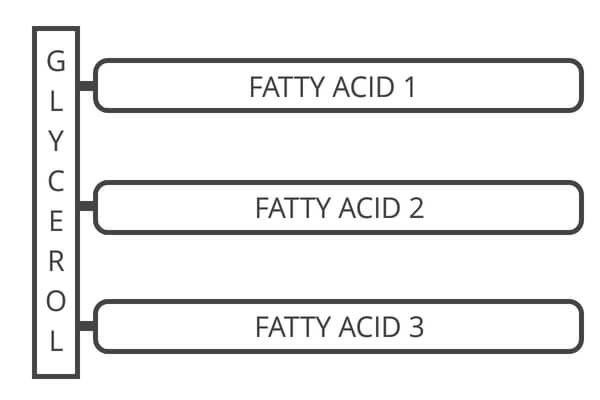
When it’s time to burn fat, your body breaks down the triglyceride into free glycerol and fatty acids.
The fatty acids circulate in the blood to the target tissue, say muscle for example.
The fatty acid enters the muscle cell and is transported to the inside of the mitochondria.
There, 2-carbon fragments at a time are cleaved off the fatty acid to create one molecule of acetyl coA.
This process is known as beta-oxidation.
This acetyl coA feeds into the Krebs cycle and is processed the very same way as glucose as described above.
Quick recap
Under normal circumstances, when you’re eating plenty of carbohydrate and fat, your body produces lots of acetyl coA from pyruvate (from glucose) or from fatty acids via beta-oxidation.
Both glycolysis and the Krebs cycle tick over unimpeded and there is peace on Earth.
However, things can go a bit haywire under fasting or starvation conditions.
I’ll explain the “carbohydrate flame” in context in the next section.
Fasting or starvation conditions
Let’s say you decide to go on the high protein, high fat pig’s feet and mayonnaise diet and your daily calorie intake is hovering around a meager 500 calories.
What happens to your glycolytic and Krebs cycle energy pathways?
First, your body uses up its stored carbohydrate (glycogen) in your liver and muscles.
Glycogen holds a bit of water too so, as you deplete your glycogen, water will go with it.
You’ll experience a bit of quick weight loss on the bathroom scale.
You’re out of glycogen, so now it’s time to start robbing Peter to pay Paul – metabolically speaking.
Second, as you’ll recall, your central nervous system prefers glucose as its primary fuel source.
If carbohydrate is not readily available, then your body can make new glucose from non-glucose sources in a process known as gluconeogenesis (gluco = glucose, neo = new, genesis = formation).
Glucose can be made from amino acids, glycerol (from triglycerides), lactate, and even oxaloacetate. (Further reading on gluconeogenesis here).
Under normal metabolism with ample glucose availability, oxaloacetate is readily available to combine with acetyl coA to form citrate which then stokes the Krebs cycle.
Oxaloacetate is normally replenished from within the Krebs cycle but can also be made from pyruvate.
But since you started the new low- or no-carb diet, you have depleted all your glycogen stores, slowed down the (glucose-fueled) Krebs cycle, and depleted your ability to make oxaloacetate from pyruvate and pyruvate from oxaloacetate.
In this particular scenario, you’re sort of boxing yourself in from both ends and creating a “metabolic traffic jam.”
Your body can send acetyl coA to the Krebs cycle (from fatty acids), but it needs to have oxaloacetate available to combine with Acetyl coA.
It is with this understanding that “fat burns in a carbohydrate flame.”
More specifically, it could be said that fat burns in an oxaloacetate flame but, since you’re unable to replenish your oxaloacetate from glucose, then your internal metabolic conditions favour the formation of ketones (or ketone bodies) for energy (ATP) production.
Ketosis and ketone formation
Glucose is your central nervous system’s preferred fuel source, but when you’re in starvation mode, gluconeogenesis is unable to produce adequate glucose from dietary carbohydrate so your body requires an alternative fuel source.
In response to either starvation (WILSON!) or a very low carbohydrate diet, hormone changes induce the breakdown of triglycerides from fat tissue and favour the formation of ketones (or ketone bodies) in your liver.
The ketone bodies are then released by your liver into the blood and are taken up and oxidised by heart, muscle, and brain tissue which contain an enzyme to convert ketones (primarily beta-hydroxybutyrate) back to acetyl coA.
Acetyl coA then feeds into the Krebs cycle to produce ATP (the “chemical currency” referred to earlier).
When you finally get rescued from the island and have a meal again (or go off of your low-carb ketogenic diet), your body will down regulate ketone production and revert back to using carbohydrate as a fuel source again.
Ketogenic diets are quite trendy and popular these days and there is evidence that they provide sustained health benefits in some people.
And, on the other hand, there are reports that lower carbohydrate dietary patterns favouring animal-derived protein and fat sources were associated with higher mortality, compared to plant-derived protein and fat sources (vegetables, nuts, grains) which were associated with lower mortality.

excellent
Many just don’t realize the OAA disappears without glycogen feeding Krebs.
Hi, what is exactly OAA and how it is made from carbs or sugars? any research papers on this?
OAA is oxaloacetate. It is made from carbohydrates (glucose/fructose/galactose)-because these 3 entities are broken down to pyruvate and pyruvate is directly converted to OAA. There is no need for you to dig up research papers because this reaction is well accepted in biochemistry and nutrition metabolism as fact (like 1 plus 1 is 2). The reaction is like this: pyruvate + CO2+biotin+pyruvate carboxylase + ATP (meaning this reaction costs you energy) yields OAA.
OAA can also be made from other sources because like the author said-glucose can be made from other sources. In fact, amino acids can produce OAA, too.
Hi. Very interesting.
What is exactly oxaloacetate? can it be only produced from sugars?
So, without ketone bodies, fatty acid oxidation is impossible? If so, is there a way to increase fat oxidation or it doesn’t matter
OAA is oxaloacetate. It is made from carbohydrates (glucose/fructose/galactose)-because these 3 entities are broken down to pyruvate and pyruvate is directly converted to OAA (and FYI, pyruvate also produces acetyl CoA). The reaction is like this: pyruvate + CO2+biotin+pyruvate carboxylase + ATP (meaning this reaction costs you energy) yields OAA.
The Krebs cycle runs to produce energy for survival-However, for this cycle to run, it must have both acetyl CoA and OAA combining together.
OAA can also be made from other sources because like the author said-glucose can be made from other sources. For instance, amino acids can produce OAA. Like wise, acetyl CoA can be formed from fatty acids, glucose metabolism (from pyruvate) and from certain amino acids.
The fatty acid unit from fat oxidation is processed further to yield acetyl CoA-this acetyl CoA, as the writer said, under normal carb consumption (v.s restricted low carb or no carb diet), enter the Krebs cycle to produce ATP to supple the body energy to live on. However, the Krebs cycle runs with the requirement of 4-carbon-units and one of those kinds is OAA from carbohydrate directly or indirect sources of carbohydrates. Because there is not enough OAA for acetyl CoA to work with in the Krebs cycle, the Krebs cycle cannot kick in and run at normal rate. Thus, the enormous amount of acetyl CoA supply from fat oxidation will not enter/be used in the Krebs cycle. But the liver comes to the rescue (to clear up the gigantic pool of acetyl CoA building up in the body.) The liver will take up 2 units of acetyl CoA and after a couple of reactions, we have acetoacetate-which can be broken down further to acetone and hydroxybutyrate. All 3 acetoacetate, hydroxybutyrate, and acetones are called ketone bodies-but mainly the acetoacetate and hydroxybutyrate play the game here. And both acetoacetate and hydroxybutyrate will be escorted by blood to tissues-here the hydroxybutyrate can be converted back to acetyl CoA and now, this acetyl CoA can enter (remember the huge pool of acetyl CoA a while ago has undergone this new pathway and taken care by the liver-and so we don’t have a bunch of acetyl CoA standing in front of the Krebs cycle yelling to get in-instead we have acceptable or workable amount of acetyl CoA at acceptable rate that the Krebs cycle with limited amount of OAA can handle and allow to enter and produce energy. Still, the rate of fat oxidation is too fast, and we have too much acetyl CoA. The liver can oxidize/metabolize acetyl CoA, but the products of this pathway by the liver produces ketone bodies-and the more acetyl CoA the liver works on, the more ketone bodies we have-and although the blood takes ketone bodies to tissues to use and convert those ketone bodies back to acetyl CoA, we simply have too much ketone bodies in the bloodstream-so while some will enter tissues and get converted to acetyl CoA to enter the Krebs cycle in “local” cells, tons of ketone bodies cannot be processed fast enough-and this condition, ketosis, is harmful defined by the human body, so the body will try to clear it up-and like other toxins, they will be eliminated via breath, sweat, feces, and urine. Yep, you bet you’ll feel and see them ketone bodies remnants in your breath and urine.
Excellent explanation step by step,although you need some basic knowledge of cell biochemistry,otherwise the reader could get easily lost.
I agree. That’s why I put in a little disclaimer at the beginning of the article. Cheers
I confess I got a little lost in there, but I’m wondering, bottom line (for me anyway), re endurance exercise when fasting and in, or approaching, ketosis, does the consumption of small (35g, say) quantities of carbs help or hinder metabolism of stored fat?
In other words, if I go on a long run and I’ve already been fasting for 48 hrs, how does the consumption of a few packs of GU (or other energy gel) impact my ability to burn stored fat?
Obviously if I consume 100kcal of carbs, you could say that’s 100kcal less of energy I’d need from my body fat given a fixed effort, but if that 100kcal of carbs essentially helps leverage my ability to burn even more fat, and exercise harder and perform (say) 300kcal more of energy expenditure than I otherwise would have been able to do, then in the end the carbs actually helped me burn fat.
So, for fat loss during endurance exercise while fasting: consume a small amount of carbs, or just water? Same Q for when not fasting.
Thanks!
Hi Peter,
The fuel you burn during exercise is not a flip-the-switch sort of thing where one second you’re burning fat and the next second you’re burning glucose. The body tends to use both of them in varying degrees.
When it comes to losing stored body fat, everyone thinks you HAVE to be burning ONLY fat as a fuel source. In general, if you’re eating different foods of differing macronutrient contents, then yes, this will have an effect on which fuel source you’re using during exercise.
But for as confusing as all the “diet gurus” out there try to make it, it still matters that you create an energy deficit. I discuss this in a bit more detail in this article: https://drbillsukala.com/fat-burn-vs-cardio-button/
Cheers
Bill
So, is a low carb, keto type diet a better way to lose fat weight? And why?
A Mediterranean style of eating is best across the board. There is a mountain of epidemiological data showing it’s excellent for weight management and health and well-being. There is also good adherence data on it. If you can eat healthy and stick with it then you’ve found the safest path to health.
Does the Krebs cycle make you lose weight? For example, taking macaroni with tomato (which has citric acid) could help burn fat?
The Krebs cycle, also called the citric acid cycle or tricarboxylic acid cycle, is only a metabolic process, not necessarily a “way to lose weight.” So eating citric acid (fruit acid) is unlikely to significantly influence the Krebs cycle in a way that would lead to appreciable weight loss.